Dr. Jackson brings many years of drug discovery and development experience to Prebys Center. Prior to joining Sanford Burnham Prebys in 2009, Dr. Jackson spent 15 years working within Johnson & Johnson’s pharmaceutical research organization. He managed cross-disciplinary teams of scientists focused on discovering chemical leads and clinical candidates directed at novel first-in-class drug targets. In 1999, he took on the role of vice president of discovery research at Johnson & Johnson’s La Jolla, California location, where he establish a state-of-the-art drugs discovery institute. As site head, Dr. Jackson oversaw all aspects of the new institute, from building design to hiring the staff of 300 scientists. In 2001 Dr. Jackson’s responsibility was expanded and as senior vice president of drug discovery at Johnson & Johnson Pharmaceutical Research and Development (U.S.) he was responsible > 800 hundred drug discovery scientists spread over five sites in the United States. Under his leadership, this organization advanced many clinical candidates into development across 5 multiple therapeutic areas including CNS, Pain, Immunology, CV disease. In 2005, Dr. Jackson was appointed President of ALZA Corporation, a large biotech company acquired by Johnson & Johnson that focused on drug delivery. As president of ALZA, he was responsible for all aspects of a 1,200 person research and development organization, successfully gaining regulatory approval for multiple drug delivery products that leverage the Oros technology and a first in class iontophoretic patch for the treatment of pain.
Prior to his industry career Dr. Jackson was an assistant professor at The Scripps Research Institute, where he published widely in the fields of cell biology and immunology. His work included identification of motifs that direct intracellular targeting of proteins to subcellular organelles, and the relevance of this and protein folding to antigen presentation and processing in the immune system. He received his PhD from the Department of Biochemistry at the University of Dundee in Scotland.
Career Highlights
- Managed (2009-2012) and then lead as P.I (2013-2014), Sanford Burnham’s NIH funded U54 grant Molecular libraries comprehensive screening center.
- Led the transformation of the Prebys center from a chemical biology and probe production organization to a full capable drug discovery operation generating first in class small molecule drug leads/NME’s.
- Instrumental in securing the Institutes first major multi-year, multi PI/project thematic translational collaboration with a pharmaceutical company.
- Instrumental in establishing the Florida Translational Research Program – a multi year contract from the department of health/State of Florida supporting Sanford Burnham Prebys translational drug discovery program in Orlando.
- Developed and executed “first of a kind” collaborative translational agreements with the Mayo clinic.
- Key member of the executive team that established a 10-year plan for the Institute that defined a path to sustainability. This plan was instrumental in securing an anonymous donation to the Institute of $275MM in January 2014.
- Established a pipeline of first in class therapeutics leads, the first product of this pipeline was licensed to Daiichi Sankyo in 2015.
- Key interface between the Institute and philanthropist Conrad Prebys, which resulted in a $100MM donation to the Institute in 2015.
- Instrumental is establishing a regional drug discovery effort for Alzheimer’s disease in collaboration with Alzheimer’s San Diego, Mayor’s office San Diego and SD County Supervisor.
Phenomena or Processes
Cell Biology
Techniques and Technologies
Drug Delivery, Drug Discovery
Dr. Jackson is senior vice president of drug discovery and development at Sanford Burnham Prebys, where he oversees the bicoastal operations of the Prebys Center, an approximately 80-person Drug Discovery enterprise embedded in the Institute. The center is equipped with state of the art ultra High Throughput Drug Screening (HTS) and lead discovery capabilities. His overall goal is to generate a pipeline of first in class small molecule therapeutics based on the break through discoveries on the molecular basis of disease made by investigators at the institute and collaborators.
Upon joining Sanford Burnham Prebys in 2009 Dr. Jackson managed and then lead as Principal Investigator, Sanford Burnham Prebys’ NIH-funded Molecular Libraries Probe comprehensive center which successfully executed over 100 HTS campaigns with collaborators across the Nation. Over a six year period the center generating >30 million screening data points and produced over 60 chemical probe reports published through the NCBI bookshelf. Since the Molecular libraries program ended the Prebys Center has continued to operate in a highly collaborative framework with investigators from many academic institutes and biopharmaceutical companies, it continues to conduct HTS screens against > 25 targets each year in search of drug leads to advance to therapeutics. Dr. Jackson is integrally involved in the center’s outreach and translational efforts that include over 35 ongoing collaborations, supported by grants, collaborations and contracts.
Dr. Jackson works closely with Sanford Burnham Prebys’ senior leadership to seek new avenues of support for preclinical and clinical drug development initiatives to enable the overall mission of the Institute to translate basic research to new products.
 Feb 12, 2025
Feb 12, 2025Curebound awards two grants to Sanford Burnham Prebys scientists
Feb 12, 2025The San Diego-based philanthropic organization has awarded $43 million in cancer research to date.
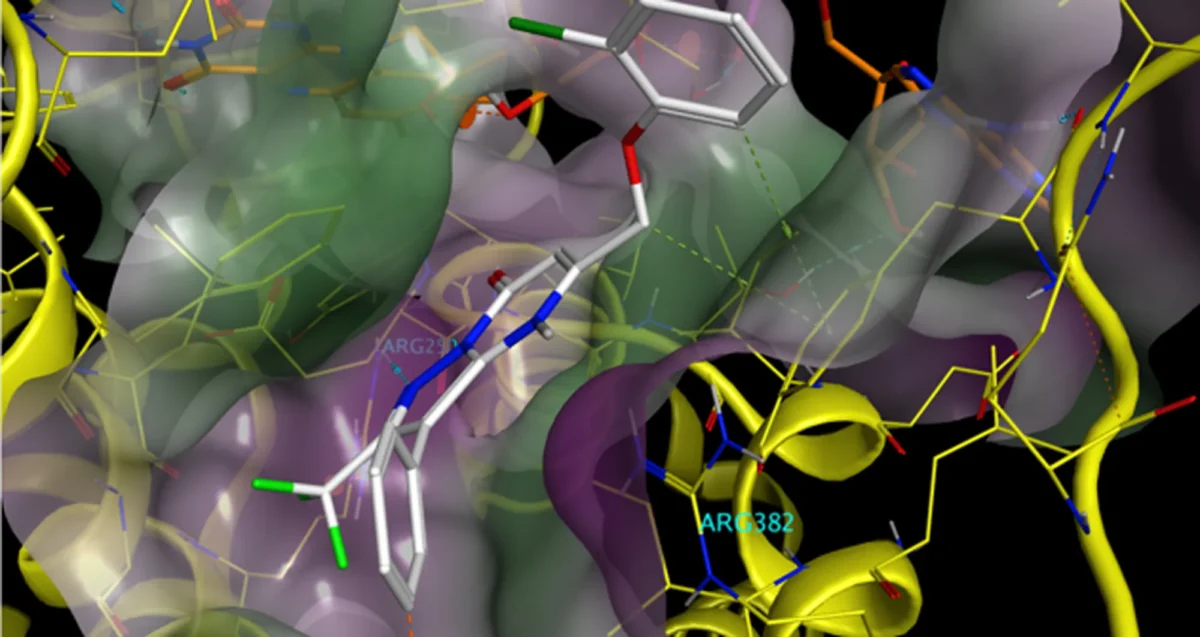 Sep 30, 2024
Sep 30, 2024How AI can make drug discovery faster, better and cheaper
Sep 30, 2024In an essay, Michael R. Jackson, PhD, senior vice president for drug discovery and development at Sanford Burnham Prebys, explains.
 Sep 5, 2024
Sep 5, 2024Acceleration by automation
Sep 5, 2024Increases in the scale and pace of research and drug discovery are being made possible by robotic automation.
 Aug 2, 2024
Aug 2, 2024Sanford Burnham Prebys event explores the science behind addiction
Aug 2, 2024Scientists and clinicians from three local research institutions converged July 31 to discuss new ways to treat multiple addictions at…
 Jul 25, 2024
Jul 25, 2024The Science Behind Addiction
Jul 25, 2024Scientists and clinicians from three local research institutions converge July 31 to discuss new ways to treat multiple addictions at…
 Nov 8, 2023
Nov 8, 2023From tobacco to alcohol to opioids, Sanford Burnham Prebys researchers are pursuing novel leads and promising therapies to treat addiction
Nov 8, 2023Addiction is perhaps the most and least visible of public health crises in the US. Tens of millions of Americans…
