Sanford Burnham Prebys scientist publishes two papers that bring us one step closer to understanding—and potentially treating—the devastating condition.
For millions of families and caregivers around the world, the need for an effective treatment for Alzheimer’s disease remains urgent despite the ongoing pandemic. Now, two studies from Timothy Huang, PhD, who was recently promoted to assistant professor in the Degenerative Diseases Program at Sanford Burnham Prebys, bring us one step closer to understanding the root cause of the disease.
Brain protein may help protect against Alzheimer’s disease
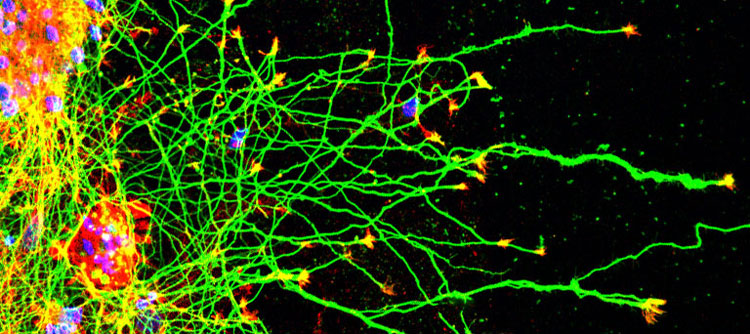
Previous research from Huang and his colleagues showed that a neuronal protein called SORLA helps reduce production of toxic amyloid beta protein that accumulates and leads to Alzheimer’s disease. Given this important role, Huang decided to dig deeper to understand SORLA’s “job” inside the brain.
In a paper featured on the cover of The Journal of Neuroscience, Huang and his team analyzed mice that produce high levels of SORLA and studied the effects of enhancing SORLA on the brain. This work showed that higher levels of SORLA resulted in elongated neurites, structures that extend from neurons, and improved the repair and regeneration of axons—the cable-like fibers that neurons use to communicate. These findings suggest that drugs that increase levels of SORLA might help protect the brain against Alzheimer’s disease and may even help people with a spinal cord injury.
Huang describes the findings as “the tip of the iceberg” and is eager to learn more about this important protein—with the ultimate goal of identifying potential targets for drugs that could slow the progression of Alzheimer’s disease.
A new model for studying Alzheimer’s disease
Many of the mutations associated with Alzheimer’s disease are found in a brain cell type called microglia. However, unlike other cells, mouse microglia are very different from human microglia. Because scientists primarily use mouse models to understand disease, this difference limits their ability to understand how microglial mutations lead to Alzheimer’s disease.
To overcome this hurdle, Huang and his team took on the formidable task of creating human stem cell lines that contain Alzheimer’s mutations found in human microglia. The scientists then tracked the downstream effects of these mutations in the cells, including epigenetic and gene expression changes, which revealed many new, previously unknown relationships between Alzheimer’s-associated genes. The findings were published in the Journal of Experimental Medicine.
More studies are needed to fully understand the how these interactions alter the course of Alzheimer’s disease—which can now be answered using this new model. Huang, who describes the work as “one of the most challenging and ambitious projects I’ve worked on so far” believes the cell line may also be used to help screen for potential Alzheimer’s disease drugs.